
Help
InverPep has at the moment 702 AMPs experimentally validated from different species and phylum of invertebrates. The peptides derived from the phylum Arthropod, Mollusca, Nematode, Annelida, Echinodermata, Platyhelminthes, Placozoa, Hydradae and the subphylum Tunichata. All the peptides are allowed in the database with their general information (phylum and species source, name, and length), the physicochemical properties (charge, hydrophobicity, Boman index, aliphatic index, pI, percentage of hydrophobic amino acids and secondary structure, if it’s known). The target organism and external link to scientific literature are also included.
CALCAMPI is a physicochemical properties calculator of multiple peptides. It was design using the programming language Perl
(version 5.22.0.1) and later it was translated to PHP to be integrated into the InverPep’s web interface. CALCAMPI calculates
molar mass, charge, hydrophobicity, Boman index, aliphatic index, isoelectric point and percentage of hydrophobic amino acids.
CALCAMPI requires a peptide list on FASTA format or an uploaded FASTA file.
- Molar mass: The peptide’s molar mass is the sum of the molar mass of all the peptide’s amino acids.
- Charge: To calculate the charge it was used the following equation:
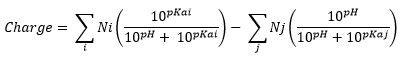
[Nelson, D., & Cox, M. (2008). Lehninger Principles of Biochemistry (5th edition ed.): W. H. Freeman.]
- Hydrophobicity: The hydrophobicity was represented in terms of the grand average of hydrophobicity. It’s calculated as the sum of the hydropathic values of each amino acid (the values are taking from the Kyte-Doolittle scale) and divided by the total number of residues. Positive values indicate hydrophobic peptides and negative values indicate hydrophilic peptides.
The percentage of hydrophobic amino acids was calculated as the sum of the hydrophobic residues (Kyte-Doolittle scale) from the peptide and divided by the total number of the residues.
[Kyte, J., & Doolittle, R. F. (1982). A simple method for displaying the hydropathic character of a protein. Journal of Molecular Biology, 157(1), 105-132.]
- Boman index: The Boman index is the sum of the free energies of the respective side chains for transfer from cyclohexane to water and divided by the total number of the residues of the peptide. The Boman index can be seen as the potencial of a peptide binding to a protein or a membrane.
[Radzicka, A., & Wolfenden, R. (1988). Comparing the polarities of the amino acids: side-chain distribution coefficients between the vapor phase, cyclohexane, 1-octanol, and neutral aqueous solution. Biochemistry, 27(5), 1664-1670.]
- Aliphatic index: The aliphatic index is the relative volume occupied for the aliphatic side chains of amino acids alanine, valine, isoleucine and leucine. Positive values are associated with thermostability of the peptide. It’s calculated with the following equation:
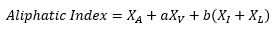
[Ikai, A. (1980). Thermostability and Aliphatic Index of Globular Proteins. Journal of Biochemistry, 88(6), 1895-1898.]
- Isoelectric point: The pI is the pH where the peptide charge is equal to zero. The pI was determined using an algorithm that calculated all the possible charges when the pH is changed, the program stops when it founds the pH value with the peptide charge equal to zero.
Blast
The Basic Local Alignment Search Tool finds regions of local similarity between sequences. The program compares nucleotide or protein sequences to sequence databases and calculates the statistical significance of matches.
http://www.rcsb.org/
[Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. (1990). Basic local alignment search tool. J. Mol. Biol. 215:403-410]
Protein Data Bank (PDB)
PDB is a crystallographic database for the three-dimensional structural data of large biological molecules, such as proteins and nucleic acids.
http://blast.ncbi.nlm.nih.gov/Blast.cgi
[H.M. Berman, J. Westbrook, Z. Feng, G. Gilliland, T.N. Bhat, H. Weissig, I.N. Shindyalov, P.E. Bourne (2000). The Protein Data Bank. Nucleic Acids Research, 28: 235-242.]
UniProt
UniProt is a space that provides a comprehensive, high-quality and freely accessible resource of protein sequence and functional information.
http://www.uniprot.org/
[The UniProt, C. (2015). UniProt: a hub for protein information. Nucleic Acids Research, 43(D1), D204-D212.]
Expasy
ExPASy is the SIB Bioinformatics Resource Portal which provides access to scientific databases and software tools (i.e., resources) in different areas of life sciences including proteomics, genomics, phylogeny, systems biology, population genetics, transcriptomics etc.
http://www.expasy.org/
[Artimo P, Jonnalagedda M, Arnold K, Baratin D, Csardi G, de Castro E, Duvaud S, Flegel V, Fortier A, Gasteiger E, Grosdidier A, Hernandez C, Ioannidis V, Kuznetsov D, Liechti R, Moretti S, Mostaguir K, Redaschi N, Rossier G, Xenarios I, and Stockinger H (2012). ExPASy: SIB bioinformatics resource portal, Nucleic Acids Res, 40(W1):W597-W603.]
HeliQuest
HeliQuest calculates from the amino acid sequence of a helix (α-helix, 3-10 helix, 3-11 helix or π helix) its physicochemical properties and amino acid composition and uses the results to screen any databank in order to identify protein segments possessing similar features.
http://heliquest.ipmc.cnrs.fr/
[Gautier R., Douguet D., Antonny B. and Drin G (2008). HELIQUEST: a web server to screen sequences with specific α-helical properties. Bioinformatics, 24(18):2101-2.]
APD
The Antimicrobial Peptide Database.
http://aps.unmc.edu/AP/main.php
[Wang, G., Li, X. and Wang, Z. (2009) APD2: the updated antimicrobial peptide database and its application in peptide design. Nucleic Acids Research 37, D933-D937.
CAMP
Collection of Anti-Microbial Peptides.
http://www.camp.bicnirrh.res.in/index.php
[Waghu FH, Barai RS, Gurung P, Idicula-Thomas S (2015). CAMPR3: a database on sequences, structures and signatures of antimicrobial peptides. Nucleic Acids Re, gkv1051v1-gkv1051.]
YADAMP
Yet another db of antimicrobial peptides.
http://yadamp.unisa.it/default.aspx
[Piotto, S. P., Sessa, L., Concilio, S., & Iannelli, P. (2012). YADAMP: yet another database of antimicrobial peptides. International Journal of Antimicrobial Agents, 39(4), 346-351.]
- Activity: Target organisms of the antimicrobial peptide. The antimicrobial activity can be specific to a single organisms
or a large group of them. E.g. Antiviral activity.
- AMP: Antimicrobial Peptides. Peptides with activity against microorganisms. E.g. Cecropins.
- InverPep code: code assigned by InverPep for each peptide in the database. E.g. 1.
- Invertebrates: organisms that don’t belong of the vertebrates’ subphylum; organisms that neither possess nor develop a
vertebral column, derived from the notochord. E.g. Mollusks.
- Length: residue´s number of the peptide. E.g. 15 amino acids.
- Peptide name: name of the antimicrobial peptide in the scientific literature. E.g. Big Defensin.
- Phylum: Phylum of the source organism of the AMP. E.g. Arthropod.
- Secondary structure: is the specific geometric shape caused by intramolecular and intermolecular hydrogen bonding of amide
groups. E.g. Alpha helix.
- Sequence: primary structure of the peptide. Letters code associated to the amino acids. E.g. ESTEVAN.
- Source species: Scientific name of the source organism of the antimicrobial peptide. E.g. Drosophila melanogaster.
You can quote InverPep using the next citation:
Gómez EA, Giraldo P, Orduz S. InverPep: a database of invertebrates antimicrobial peptides. J Glob Antimicrob Resist 2017; 8:13-17.
Thank your for wanting to contribute with InverPep. To upload new peptides you can follow the next link and complete the cuestionary: Cuestionary
All the InverPep's peptides had been experimentally validated and in the information pages of each one you can find a link to the scientific literature that contains this comprobation
Contacts
Grupo de Biología Funcional
Research's area of Ecología y Sistemática de Insectos
Facultad de Ciencias
Universidad Nacional de Colombia - Sede Medellín
Contact
Sergio Orduz, Biol., M.Sc., Ph.D.
Coordinator of Grupo de Biología Funcional
sorduzp@unal.edu.co / 4306395 / Laboratory of Prospección y Diseño de Biomoléculas (Bl 16 - 211)
Participants
InverPep
| Creation and manual annotation | CALCAMPI | Interface design | Relization web database |
|---|---|---|---|
| Esteban Gómez Paula Andrea Giraldo |
Esteban Gómez | Esteban Gómez Juan David Cadavid Q. Ricardo Mesa F. |
Juan David Cadavid Q. Ricardo Mesa F. Esteban Gómez |


